Although mandatory vaccine campaigns went relatively smoothly in the developing world, there were sporadic reports of detecting fake vaccine certificates or fake virus tests to evade vaccination. 
Recently, with the severe surge of COVID-19 cases and emergence of aggressive virus variants several countries such as the United States, Italy, and France have mandated vaccines starting from July 2021, 7 while other countries announced that they will start mandatory vaccination early 2022. To increase vaccination rates several countries mandated vaccination for eligible candidates as soon as vaccines were approved. 
Moreover, vaccine hesitancy was reported among health-care workers who demonstrated unacceptably low vaccination rates at times. Despite efforts of the scientific community to assure the public about the safety and efficacy of vaccines, there has been significant public hesitancy toward COVID-19 vaccination. Public trust in vaccines has been influenced by several political and social factors with concerns over the relatively rapid production, and claims of inadequate testing and monitoring of vaccine safety. Potential side effects of vaccines, and concerns about their safety profile represent important obstacles against efforts to distribute vaccines among eligible candidates. 5 Side effects were described as local effects in the injection site and systemic transient manifestations in the form of fatigue, headache, fever, among others. 1–4Įarly reports on these vaccines concluded that their side effects are mostly acute in nature, with a duration ranging from 1 to 10 days. These vaccines have been reported to have efficacy rates ranging from 79% to 95% in reducing risk of moderate to severe illness, hospitalization, and deaths related to COVID-19. Several vaccines (utilizing three main platforms) were approved and used globally, namely Pfizer-BioNtech (PB) (RNA-based), AstraZeneca (AZ) (Adenoviral vector-based), and Sinopharm (SF) (inactivated virus). On the other hand, Sinopharm vaccine showed the highest significant association with these AEs followed by AstraZeneca vaccine.Ī year has passed since the roll out of Corona virus disease-2019 (COVID-19) vaccines. It seems that factors such as age, gender, and medical status play a negligible role in development of these AEs. Less than 20% of COVID-19 vaccine recipients may complain of LTAEs that are mostly fatigue-related. This was further confirmed by general linear multivariate model analysis. The collective symptoms of fatigue, myalgia, arthralgia, dizziness, and headache were significantly associated with Sinopharm vaccine ( P = .04). There was no statistically significant association between LTAEs and: age, gender, or medical history ( P > .05). A total of 80 (16.0%) participants reported LTAEs which were mainly fatigue, menstrual disturbances, myalgia, arthralgia, dizziness, and headache (N = 32, 15, 8, 6, 4, and 4, respectively). The most frequently administered vaccines were Pfizer-BioNtech, Sinopharm and AstraZeneca vaccines. 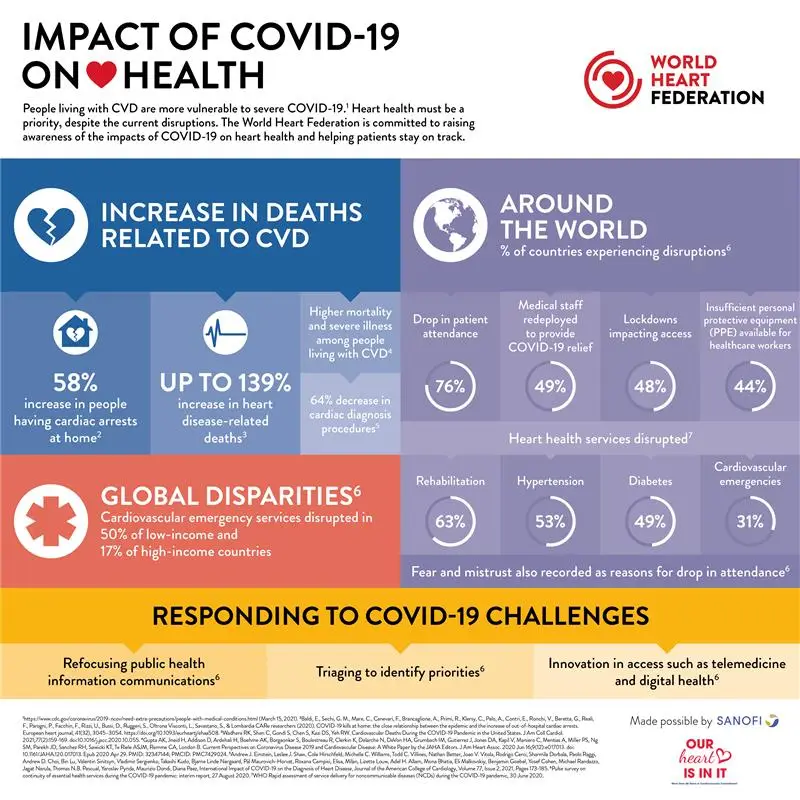
Data of a total of 498 practitioners were included. A group of physicians and dentists were invited to complete a validated questionnaire that was composed of items on: socio-demographics, medical history, administered vaccines, and long-term adverse events (LTAE). This study aims to explore long-term adverse events reported by physicians and dentists who received at least two COVID-19 vaccine doses. Assessment of safety of COVID-19 vaccines is an ongoing process. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
